Updated on October 17, 2023
In China, health food is usually defined as food products that have specific health functions or supply vitamins and (or) minerals. With the goal of regulating the body's function, health food is suitable for specific groups of people. However, it is not used for the purpose of curing disease and causes no acute, sub-acute or chronic health effect to human body.
The Definitions/Names of these Food Products in Different Countries are Different:
- China: Health Food
- EU: Food Supplement
- USA: Dietary Supplement
- Canada: Natural Health Product
- Australia: Complementary Medicines
- Korea: Health Functional Food
- Japan: Food with Health Claims (FHC)
Classification of Health Food
I. Nutrition supplement:
Food that replenishes vitamins, minerals and other nutrients but without providing energy or other active ingredients.
II. Functional health food:
Food that labeled with health function claim to have physiological effects on the human body.
Regulation Background
In accordance with Food Safety Law of the People’s Republic of China (2015 version), companies who plan to place health food in the Chinese market shall apply for and obtain the health food registration certificate or filing certificate. For domestic health foods produced in China, the registration shall be conducted with the State Administration for Market Regulation (SAMR, former CFDA), whereas, the filing shall be carried out with the Provincial Administration for Market Regulation. For imported health foods produced in overseas factories, both the registration and filing shall be applied with SAMR. Meanwhile, overseas companies shall have a permanent Chinese representative office or appoint a Chinese agent to deal with registration or filing and obtain such certificates.
News Updates
In accordance with the announcement of the State Administration for Market Regulation (SAMR) regarding the release of Directory of Health Functions Available to Be Claimed by Health Food - Non-nutrition Supplements (2023 Version) and the supporting documents, registrants need to change their health food registration certificates to revise the wording of their health claims, so as to align the latest wording and evaluation requirements when the health functions are listed in the Directory yet the wording of the health claims is outdated (e.g., regulating immunity or improving sleep).
Relevant Regulations on Health Food
Name | Released Date | Implemented Date |
Food Safety Law of the People's Republic of China (2021 Revised Version) | 2015.04.24 (Revised on 2021.04.29) | 2015.10.01 |
Administrative Measure on Health Food Registration and Filing (2020 Revised Version) | 2016.02.26 (Revised on 2020.11.03) | 2016.07.01 |
2014.12.24 | 2015.05.24 | |
Health Food Registration Review Rules (2016 Version) | 2016.11.14 | 2016.11.14 |
Health Food Registration Application Service Guideline (2016 Version) | 2016.12.19 | 2016.12.19 |
2019.08.20 | 2020.01.01 | |
Guidelines for Naming of Health Food (2019 Version) | 2019.11.12 | 2019.11.12 |
Guideline on the Safety Toxicological Inspection and Evaluation of Health Food and Its Raw Materials (2020 Version) | 2020.10.31 | 2020.10.31 |
Guideline on the Safety Inspection and Evaluation of Strains Used in Health Food Raw Materials (2020 Version) | 2020.10.31 | 2020.10.31 |
Guideline on the Inspection and Evaluation of Physicochemical and Hygienic Indicators of Health Food (2020 Version) | 2020.10.31 | 2020.10.31 |
2023.08.31 | 2023.08.31 | |
Testing and Evaluation Methods for Health Food Functions (2023 Version) | 2023.08.31 | 2023.08.31 |
Guideline on Testing and Evaluation of Health Food Functions (2023 Version) | 2023.08.31 | 2023.08.31 |
Guideline on Ethical Review of Health Food Human Feeding Trials (2023 Version) | 2023.08.31 | 2023.08.31 |
Health Food Filing Guideline (Trial) | 2017.05.02 | 2017.05.02 |
Health Food Raw Materials Directory of Nutrition Supplement (2023 version) | 2023.06.14 | 2023.10.01 |
Health Function Directory of Allowing Nutrition Supplement Claims (2023 version) | 2023.06.14 | 2023.10.01 |
Health Food Raw Materials Directory of Soy Protein Isolate | 2023.06.14 | 2023.10.01 |
Health Food Raw Materials Directory of Whey Protein | 2023.06.14 | 2023.10.01 |
Dosage Forms and Technical Requirements of Filing Product with Health Food Raw Material (Soy Protein Isolate, Whey Protein) | 2023.09.29 | 2023.10.01 |
Health Food Raw Material Directory of Coenzyme Q10, Melatonin, Fish oil, Broken Ganoderma lucidum spore powder and Spirulina | 2020.12.01 | 2021.03.01 |
Available Excipients for Health Food Filing and Their Usage Rules (2021 version) | 2021.02.20 | 2021.06.01 |
Dosage Forms and Technical Requirements of Health Food Filing (2021 version) | 2021.02.20 | 2021.06.01 |
Health food registration and filing shall be carried out according to the Administrative Measure on Health Food Registration and Filing (herein named the Measure). The Detailed information are as follows:
Part I Health Food Filing
(People like to call it “Red/Orange Hat”, in fact, once the filing certificate is received, the logo of “Blue Hat” rather than “Red/Orange Hat” will be put on the label)
Applicable Scope of Filing
Nutrition Supplement: The health food whose formulation meets the requirements in Health Food Raw Materials Directory and Available Excipients for Health Food Filing and Their Usage Rules (Trial) can do the filing rather than registration.
Applicant's Qualification of Filing
I. The filing applicant of domestic health food shall be the factory that has the production certificate. In other words, the domestic applicant cannot entrust production.
II. The filing applicant of imported health food could be the overseas manufacturer (overseas manufacturer refers to the legal person and other organization). In other words, the overseas applicant could entrust production.
Filing Procedure
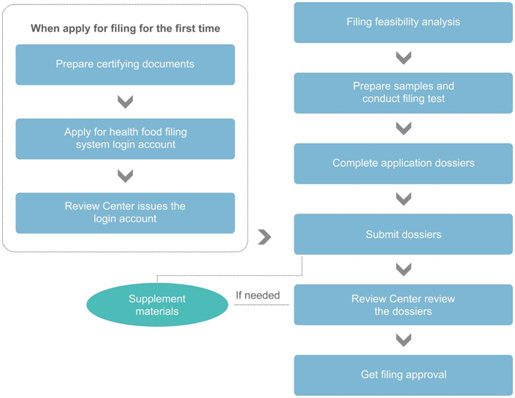
Dossier Requirements for Health Food Filing System Login Account Application
If it is the first time to apply for filing, the applicant shall get the health food filing system login account in advance. The required documents are as following:
- Basic company information and contact information;
- Qualification certifying document of the applicant;
- Letter of Authorization of the contact;
- Scanned passport/ identification card of legal representative.
Dossier Requirements of Filing
According to the Measure, the following documents are required for health food filing:
- Health food filing application form; Letter of commitment for authenticity of the materials;
- Copies of legally registered certificates of the applicant;
- Product formulation materials (APIs and excipients);
- Product production process materials;
- Stability test report, and the explanation of the use of active ingredients and excipients;
- Information of packaging materials in direct contact with the product;
- Samples of product label and package insert;
- Product technical requirements;
- Test according to product technical requirements;
- Other materials prove product safety and health function.
For imported health food filing, besides the above documents, the following supplementary documents also should be submitted:
- Qualification certifying documents issued by government authorities or legal service agencies in the producing country (region) of origin proving that the overseas filing applicant is the owner of the health food marketed;
- Certifying documents issued by government authorities or legal service agencies in the producing country (region) of origin proving that the product has been marketed more than a year, or safety report of overseas sales and consumer’s feedback;
- Health food-associated standards issued by the product-producing country (region) of origin or international organizations;
- Packaging, labels, and package inserts for products marketed in the producing country (region)of origin;
- For filing affairs run by overseas manufacturer’s Permanent Representative in China, a copy of the "registration certificate of overseas enterprise’s permanent Chinese representative offices" shall be provided; for filing affairs run by domestic agencies entrusted by oversea manufacturers, the applicant shall provide the original notarized certificate of entrustment and copies of business license of the agencies entrusted.
Test Requirements of Filing
According to the Measure, the following tests are required to be arranged in CFDA designated testing institutions for health food filing:
- Tests listed in the document of technical requirements including Functional components/characteristic ingredients test, Hygiene health test, etc. for three batches of samples;
- Stability test for three batches of samples.
More details on the health food testing could be found here.
Part II Health Food Registration
(Once the registration certificate is received, the logo of “Blue Hat” can be put on the label)
Applicable Scope of Registration
Functional Health Food: The health food whose active ingredients are out of the scope of the Health Food Raw Material Directory. And it has some specific health functions for specific groups of people.
24 Health Claims Available for Functional Health Foods
Earlier in August 2023, China finally released the Directory of Health Functions Available to be Claimed by Health Food – Non-nutrition Supplements (2023 Version) and the supporting documents, including the long-awaited health food function evaluation methods, in which the total number of health functions has been reduced from 27 to 24.
S.N. | Health functions |
1 | Aids in enhancing immunity |
2 | Aids in anti-oxidation |
3 | Aids in improving memory |
4 | Alleviating visual fatigue |
5 | Soothes and moistens the throat |
6 | Aids in improving sleep |
7 | Alleviating physical fatigue |
8 | Tolerant to hypoxia |
9 | Aids in controlling body fats |
10 | Aids in increasing bone density |
11 | Improving iron-deficiency anemia |
12 | Aids in eliminating acne |
13 | Aids in eliminating skin chloasma |
14 | Aids in improving skin moisture condition |
15 | Aids in regulating intestinal microbiota |
16 | Aids in promoting digestion |
17 | Aids in promoting regular bowel movements |
18 | Aids in protecting the gastric mucosa |
19 | Aids in maintaining healthy blood lipid (cholesterol/triglyceride) levels |
20 | Aids in maintaining healthy blood sugar levels |
21 | Aids in maintaining healthy blood pressure levels |
22 | Providing auxiliary protective action against chemical liver damage |
23 | Provides auxiliary protective action against ionizing radiation hazards |
24 | Aids in lead excretion |
Applicant's Qualification of Registration
I. The registration applicant of domestic health food could be the legal person or other organizations registered in China. In other words, the domestic registration applicant could entrust production.
II. The registration applicant of Imported health food could be the overseas manufacturer (overseas manufacturer refers to the legal person and other organization). In other words, the overseas registration applicant could entrust production.
Registration Procedure
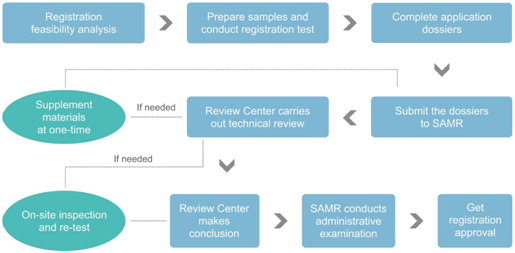
Dossier Requirements of Registration
According to the Measure, the following documents are required for health food registration:
- Health food registration application form; Letter of commitment for the authenticity of the materials;
- Copies of legally registered certificates of the applicant;
- Product development report including the product technical requirements;
- Product formulation materials (APIs and excipients);
- Product production process materials;
- Safety and function assessment material; and tests reports of functional components/characteristic ingredients, stability, hygiene health and others if necessary;
- Information of packaging materials in direct contact with the product;
- Samples of product label and package insert;
- 3 samples with the minimum sales packaging;
- Other materials pertaining to the product registration technical evaluation.
For imported health food registration, besides the above documents, the following supplementary documents also should be submitted:
- Qualification certifying documents issued by government authorities or legal service agencies in the producing country (region) of origin proving that the overseas registration applicant is the owner of the health food marketed;
- Certifying documents issued by government authorities or legal service agencies in the producing country (region) of origin proving that the product has been marketed more than a year, or safety report of overseas sales and consumer’s feedback;
- Health food-associated standards issued by the product-producing country (region) of origin or international organizations;
- Packaging, labels, and package inserts for products marketed in the producing country (region)of origin;
- For registration affairs run by an overseas manufacturer’s Permanent Representative in China, a copy of the "registration certificate of overseas enterprise’s permanent Chinese representative offices" shall be provided; for registration affairs run by domestic agencies entrusted by oversea manufacturers, the applicant shall provide the original notarized certificate of entrustment and copies of business license of the agencies entrusted.
Test Requirements of Registration
According to the Measure, the following tests are required to be arranged in CFDA-designated testing institutions for health food registration:
- Safety and toxicology test;
- Animal and (or) human function test;
- Functional components/characteristic ingredients test, Hygiene health test;
- Stability test;
- Strain identification and strain virulence test for Probiotics-based Health Food;
- Stimulants, illicit drugs test for Health Food with Relieving physical fatigue, Weight loss, or Improving growth and development function;
- Other tests if necessary
More details on the health food testing could be found here.
Our Services
CIRS provides one-stop services for China food regulatory compliance. We also deliver the most up-to-date regulatory information about food safety control in China. For health food, we offer the following services:
I. Training on Health Food Regulation
II. Health Food Regulation Update Monitoring
III. Health Food Registration
IV. Health Food Filing
V. Single Technology Services
- Pre-market Investigation
- Classification Analysis and Formula Review
- Chinese Label and Package Insert Design
- Dossier Preparation
- Translation
- Test Arrange and Monitoring
If you need any assistance or have any questions, please get in touch with us via service@cirs-group.com.
Further Information
Registration Status of Health Food in China in the first half of 2023
Filing Status of Chinese Health Foods (Dietary Supplements) in the First Half of 2023
Analysis on the Filing Status of Health Food (Dietary Supplement) in 2023 in China

